TMS-induced modulation of brain networks and its associations to rTMS treatment for depression: a concurrent fMRI-EEG-TMS study
He, H., Sun, X., Doose, J., Faller, J., McIntosh, J. R., Saber, G. T., ... & Sajda, P. (2025). TMS-induced modulation of brain networks and its associations to rTMS treatment for depression: a concurrent fMRI-EEG-TMS study. Brain Stimulation.
Introduction
This study investigates the immediate effects of transcranial magnetic stimulation (TMS) on brain networks and their relationship to treatment outcomes in patients with treatment-resistant depression (TRD). Using a novel, integrative approach that simultaneously combines TMS with functional magnetic resonance imaging (fMRI) and electroencephalography (EEG), the researchers aimed to understand the therapeutic mechanisms of TMS, particularly how the brain's state at the moment of stimulation influences its effects. The work demonstrates that TMS acutely modulates critical brain networks and that these modulations, when timed to a patient's specific brain rhythms, are associated with clinical improvement.
Research Question
While TMS over the left dorsolateral prefrontal cortex (L-DLPFC) is an FDA-approved treatment for depression, its underlying mechanisms are not fully understood. It is hypothesized that the therapeutic effects depend not only on stimulating the correct location but also on the timing of the stimulation relative to the brain's ongoing oscillatory state. This study sought to test this hypothesis by exploring whether synchronizing TMS pulses with a patient's prefrontal alpha-wave oscillations could enhance target engagement and predict treatment response, thereby paving the way for more personalized and effective neuromodulation therapies.
Methodology
This research was a randomized, double-blind clinical trial involving 28 patients with TRD. The methodology can be broken down into three main phases:
Baseline Assessment
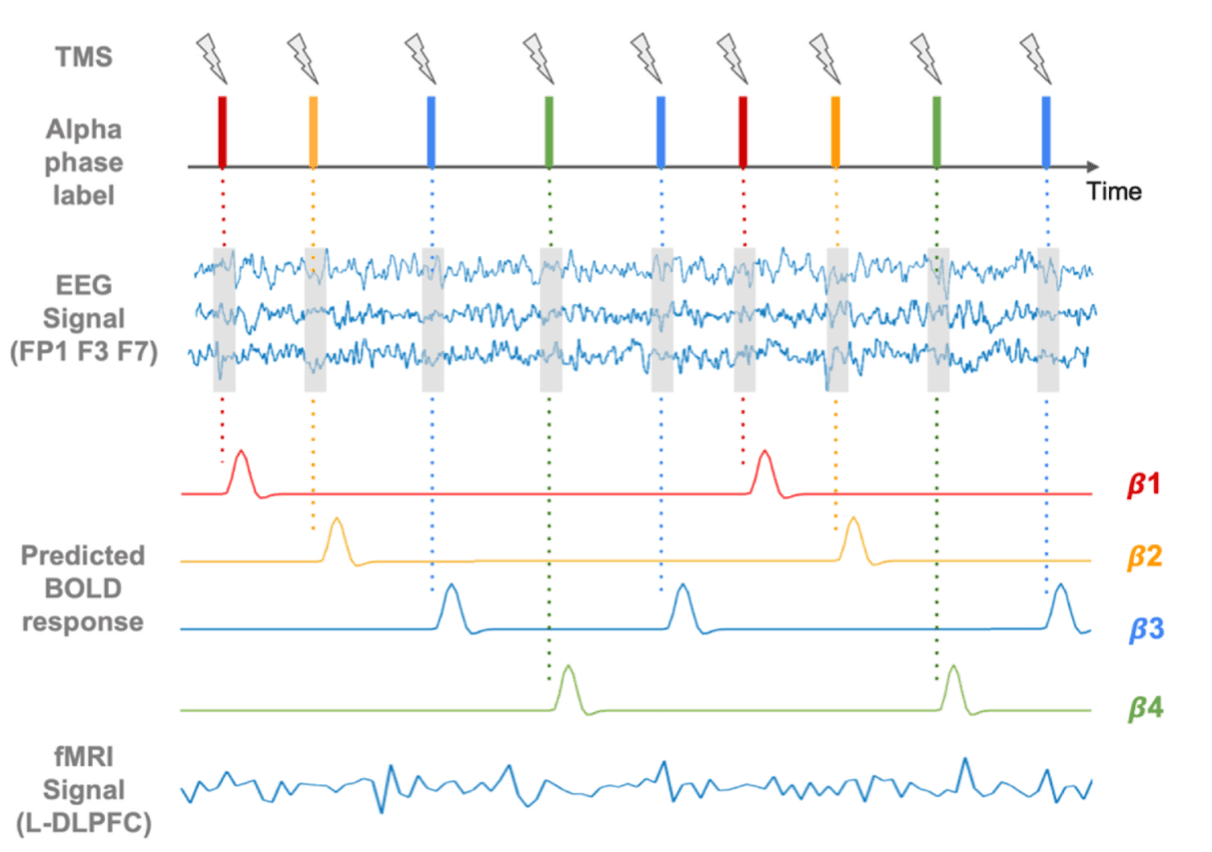
Each patient underwent a pre-treatment scan using an integrated fMRI-EEG-TMS (fET) instrument. During this scan, single pulses of TMS were delivered to the L-DLPFC. The concurrent EEG data was used to identify a personalized "optimal phase" of the prefrontal alpha oscillation—the specific point in the brainwave cycle where TMS produced the largest BOLD signal increase in the dorsal anterior cingulate cortex (dACC), a key region in mood regulation.
rTMS Treatment
Patients were randomized into two groups for a six-week treatment protocol.
SYNC Group (n=15): Received repetitive TMS (rTMS) pulses synchronized to their individually determined optimal alpha phase.
UNSYNC Group (n=13): Received rTMS pulses at the same frequency but at random phases.
Data Analysis
The researchers analyzed the acute effects of single-pulse TMS from the pre- and post-treatment fET scans. They quantified TMS-evoked BOLD responses across the brain and assessed TMS-induced changes in functional connectivity using a psychophysiological interaction (PPI) analysis. Brain state was defined by the phase of the EEG prefrontal alpha oscillation at the time of each TMS pulse, allowing for state-dependent analysis. Clinical outcomes were measured by the percent change in the Hamilton Rating Scale for Depression (HRSD).
Key Findings
The study yielded several key insights into the mechanisms and predictors of TMS efficacy:
Acute Network Modulation
Single-pulse TMS was found to acutely modulate connectivity within and between large-scale brain networks, particularly by decreasing connectivity involving the default mode network (DMN), cognitive control network (CCN), and limbic networks.
Predicting Treatment Response
For patients in the SYNC group only, the strength of the baseline TMS-evoked BOLD response was a powerful predictor of clinical improvement. Specifically, stronger responses in the bilateral cognitive control network (r ≈ 0.86) and the right limbic network (r ≈ 0.90) were highly correlated with greater symptom reduction. This association was not present in the UNSYNC group.
State-Dependent Effects
The study confirmed that the effects of TMS are brain-state dependent. TMS pulses delivered during a "high-load phase" (the phase that evoked the strongest response at the stimulation site) produced significant downstream network modulation, whereas pulses at a "low-load phase" did not.


Circuit-Specific Changes
The clinical improvement in the SYNC group was strongly associated with longitudinal changes in state-specific functional connectivity. A greater increase in connectivity modulation between the L-DLPFC and the right orbitofrontal cortex (a part of the subgenual cingulate circuit) from pre- to post-treatment was almost perfectly correlated with a better clinical outcome (r = 0.99).

Implications
This study provides compelling evidence for the mechanisms underlying TMS treatment for depression, emphasizing the importance of personalized timing for optimizing target engagement. The findings suggest that EEG-synchronized rTMS may work by systematically engaging a specific and clinically relevant brain circuit connecting the L-DLPFC and the subgenual cingulate cortex.
The ability of baseline TMS-evoked responses to predict outcomes in the SYNC group offers a potential biomarker for identifying patients who would benefit most from this personalized approach. However, the authors acknowledge several limitations, including a small sample size and the absence of a sham control group, which means placebo effects cannot be ruled out. Furthermore, the clinical trial did not find a significant difference in overall efficacy between the SYNC and UNSYNC groups. Despite this, the neuroimaging results strongly support the mechanistic role of state-dependent circuit engagement and are crucial for guiding future research into developing more precise and personalized neuromodulation therapies for TRD.